Obesity threatens to shorten the human lifespan by 5–20 years and refers to many aspects of biology ,rather than that was previously appreciated, What’s worse is the obesity-induced diseases. Currently, therapy for curing obesity is limited by fully understanding of the mechanism for controlling obesity.
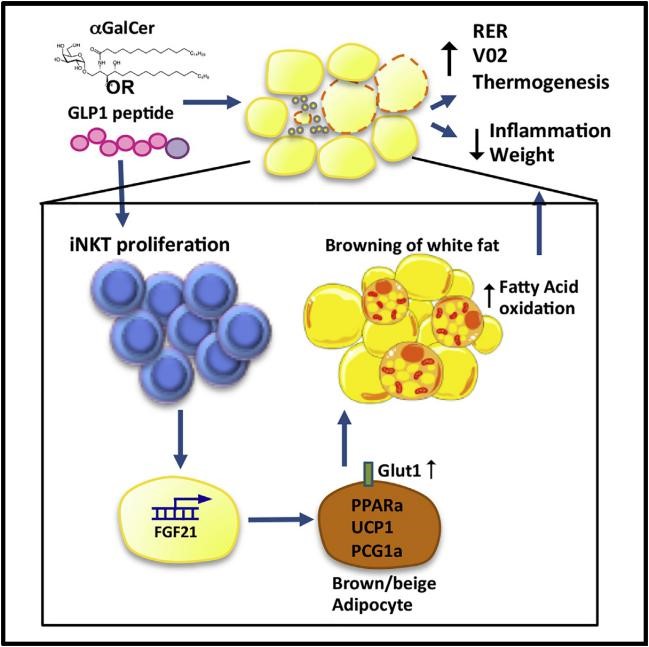
A promising potential approach for treating obesity is activation of brown adipose tissue (BAT). In contrast to energy-storing white adipose tissue (WAT), BAT contains many thermogenic mitochondria which secretes uncoupling protein 1 (UCP-1) that converted chemical energy into heat, Recent studies have defined the role of the adipose innate immune system in the regulation of metabolism and control of body weight, Invariant natural killer T ( iNKT ) cells are one such innate immune cell type with an important role in weight and glycemic control.
iNKT cells are key player in metabolic regulation, iNKT cells are innate lipid sensors , using their prototypic ligand a-galactosylceramide (aGalCer) to being activtion can lead to weight loss and glycemic control. Here, iNKT activation induced fibroblast growth factor 21 (FGF21) production and thermo-genic browning of white fat. Complete metabolic analysis revealed that iNKT cell activation induced increased body temperature and fatty acid oxidation, without affecting food intake or activity. FGF21 induction played a major role in iNKT cell-induced weight loss, as FGF21 null mice lost significantly less weight after aGalCer treatment. The glucagon-like peptide 1 (GLP-1) receptor agonist-liraglutide also activated iNKT cells in humans and mice. In iNKT-deficient mice, liraglutide promoted satiety but failed to induce FGF21, which resulting in less weight loss. These findings indicates an iNKT cell-FGF21 axis may defines a new immune-mediated pathway that could be regulate for glycemic control and weight regulation.